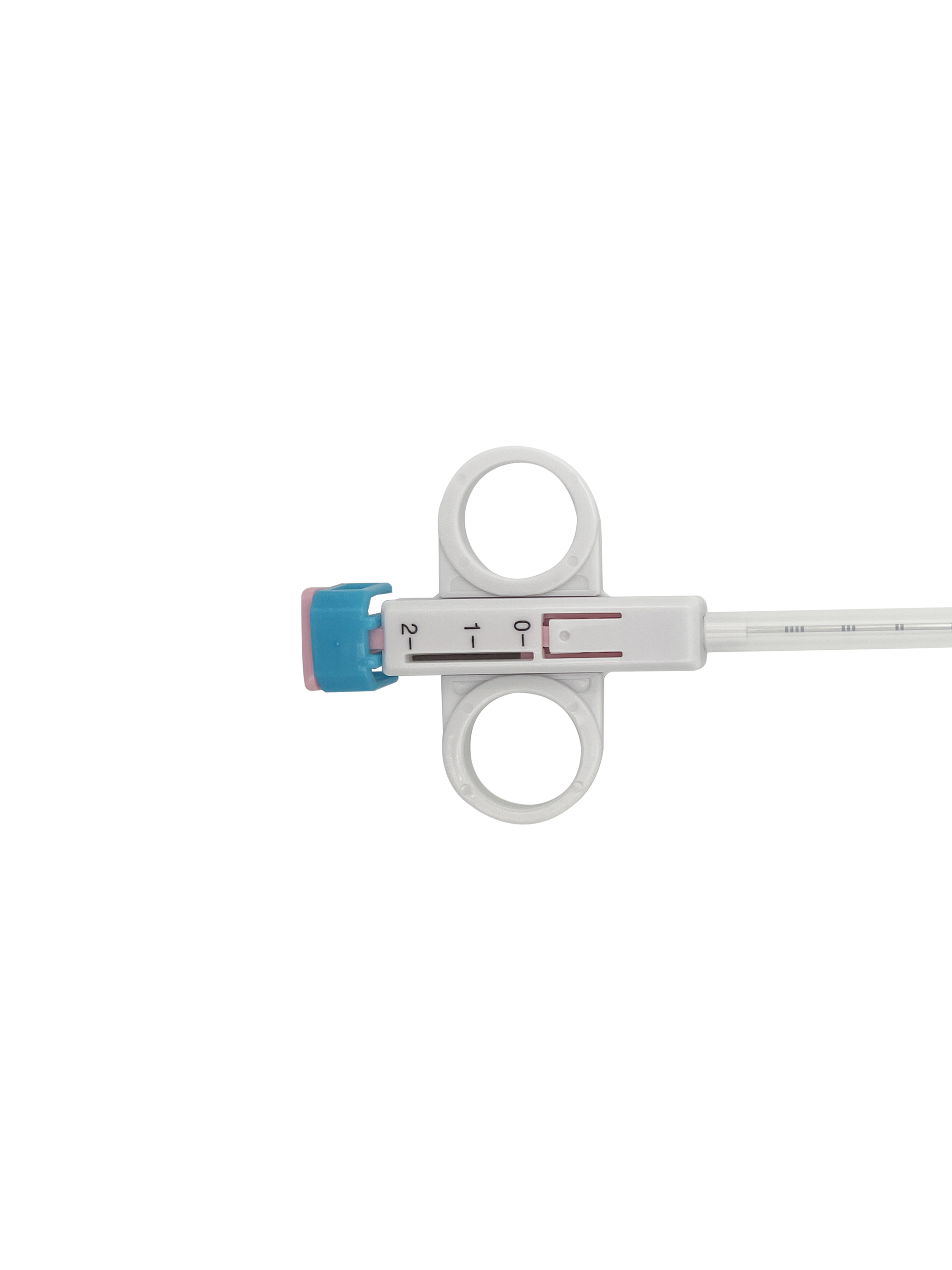
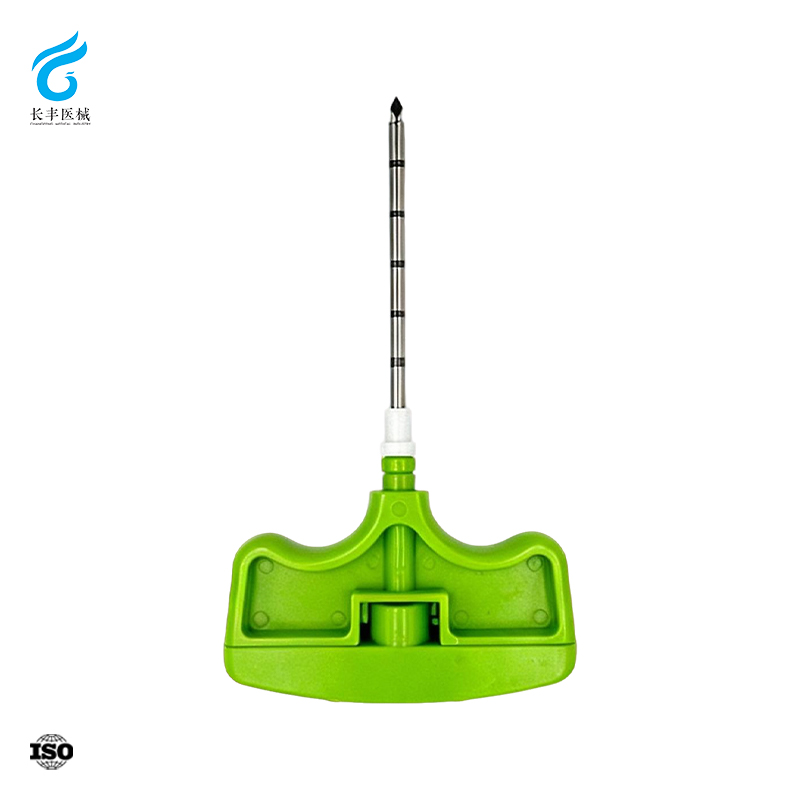
La gestión de la calidad y el cumplimiento normativo son fundamentales para el rendimiento de las pistolas automáticas de biopsia, la confianza clínica y el acceso a los mercados globales. Jiangsu Changfeng Medical Industry Co., Ltd. mantiene sistemas integrales de calidad que abarcan el diseño, la adquisición de materiales, la producción, la esterilización, el embalaje y la distribución de las pistolas automáticas de biopsia. Nuestros procesos de fabricación cumplen con las normativas internacionales aplicables a los dispositivos médicos, sometiendo cada producto a ensayos rigurosos de precisión dimensional, agudeza de la aguja, consistencia en el disparo, estanqueidad, esterilidad y biocompatibilidad. Implementamos una trazabilidad completa por lotes a lo largo de toda la cadena, garantizando transparencia desde las materias primas hasta los usuarios finales. Modernas líneas de mecanizado de precisión ubicadas en Yangzhou permiten una fabricación consistente y escalable, manteniendo al mismo tiempo estrictos estándares de calidad. Nuestros centros de I+D en Hangzhou y Suzhou impulsan de forma continua la mejora del diseño, los materiales y los procesos para satisfacer los requisitos normativos y clínicos en constante evolución. Las pistolas automáticas de biopsia de Changfeng son adecuadas para licitaciones hospitalarias, asociaciones con distribuidores y exportación internacional, respaldadas por expedientes técnicos completos, certificados y etiquetado conforme a la normativa. Priorizamos la gestión de riesgos durante todo el ciclo de vida del producto para minimizar peligros y garantizar una fiabilidad a largo plazo. Como empresa médica moderna integrada, defendemos una producción ética, la responsabilidad medioambiental y una gobernanza corporativa sólida. Nuestra alineación con la estrategia «China Saludable» refuerza nuestro compromiso con dispositivos médicos seguros, regulados y accesibles. Para obtener información detallada sobre certificaciones de calidad, documentos de cumplimiento normativo, informes de auditoría y precios de nuestras pistolas automáticas de biopsia, póngase en contacto con nuestro equipo de calidad y ventas.